|
8/23/2023 0 Comments Increase in entropy
It is never less than the change of heat of a system per unit degree of temperature. Note:It is to be noted that the entropy is always greater than the change of heat of system per unit degree of temperature for an irreversible process while the entropy is always equal to the change of heat of system per unit degree of temperature for a reversible process. Since entropy is a point function that depends only on states i.e. It seems that the entropy of the universe is only increasing. This can be seen in Figure 18.3 Entropy vs. No process between two equilibrium states is possible if it would result in a decrease in the total entropy of the system and surroundings. The idea of the irreversible process is a critical one for entropy change in the second law. The standard molar entropy of any substance increases as the temperature increases. 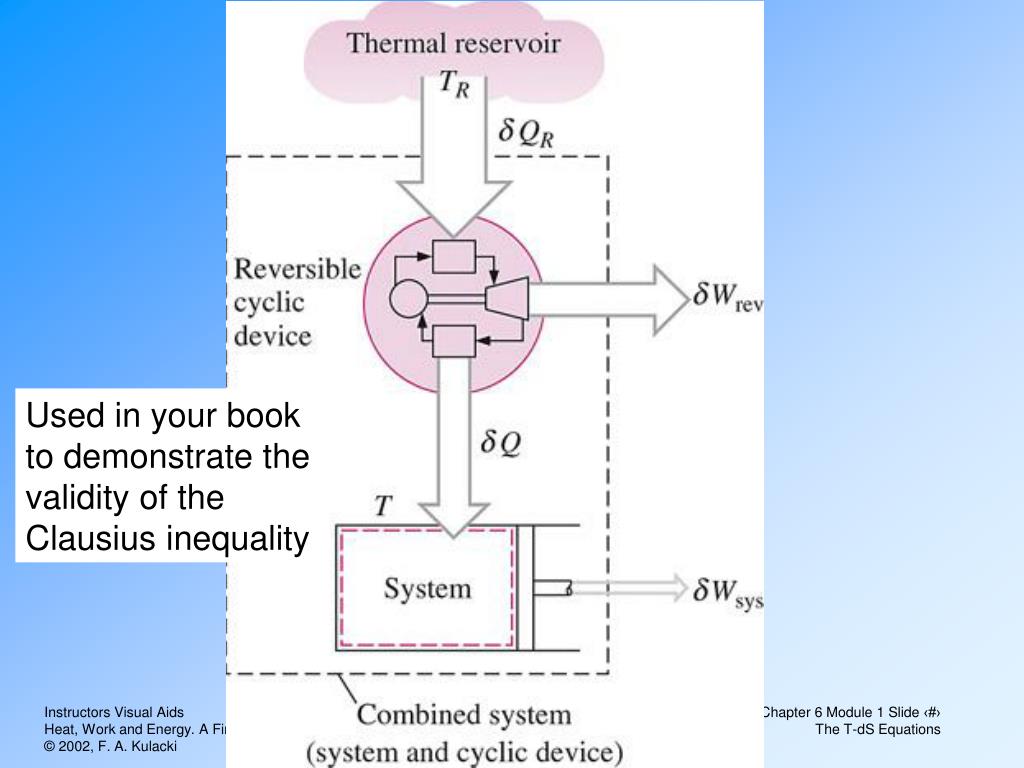
Therefore, the entropy of an isolated system either increases or in the limit, remains constant. Increase in entropy of a system leads to increase in randomness and degradation of energy. From them we can draw some general conclusions. 
The entropy change of a system is given by the equation: Below we have various examples of entropy change during various processes. It never decreases but remains constant only in a reversible process. The principle of increase of entropy states that the entropy of the Universe also increases. Where, \is the change of heat of the system and \is the temperature of the system.Įntropy is defined as the property of the randomness or disorder in a system. Hint:To solve this question, we are going to firstly define the entropy of a system then, the principle of entropy is defined after which the inequality and equality relations for the entropy are given along with their specification with the reversible or irreversible nature of the process of system.įormula used: The entropy change of a system is given by the equation:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
